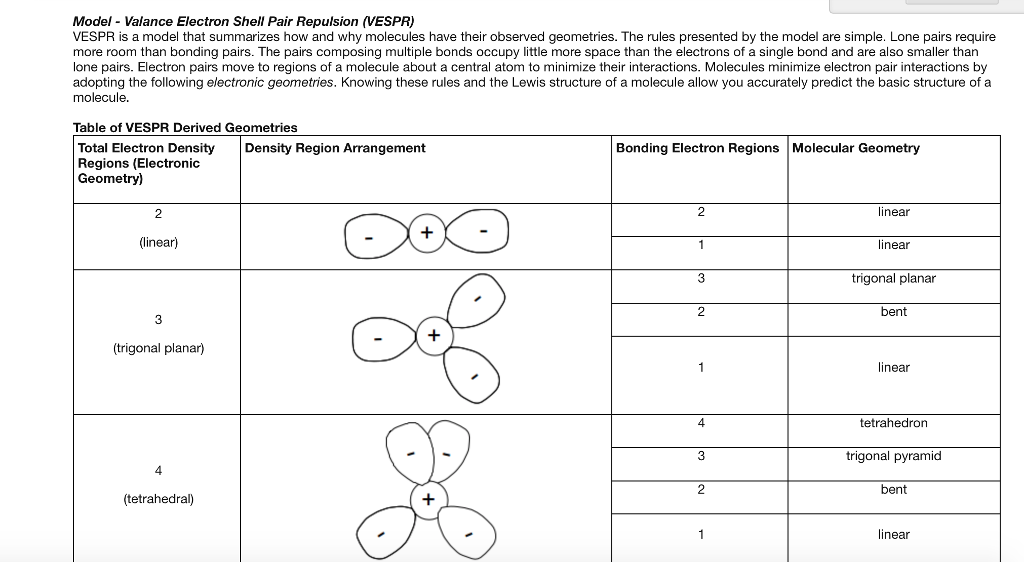  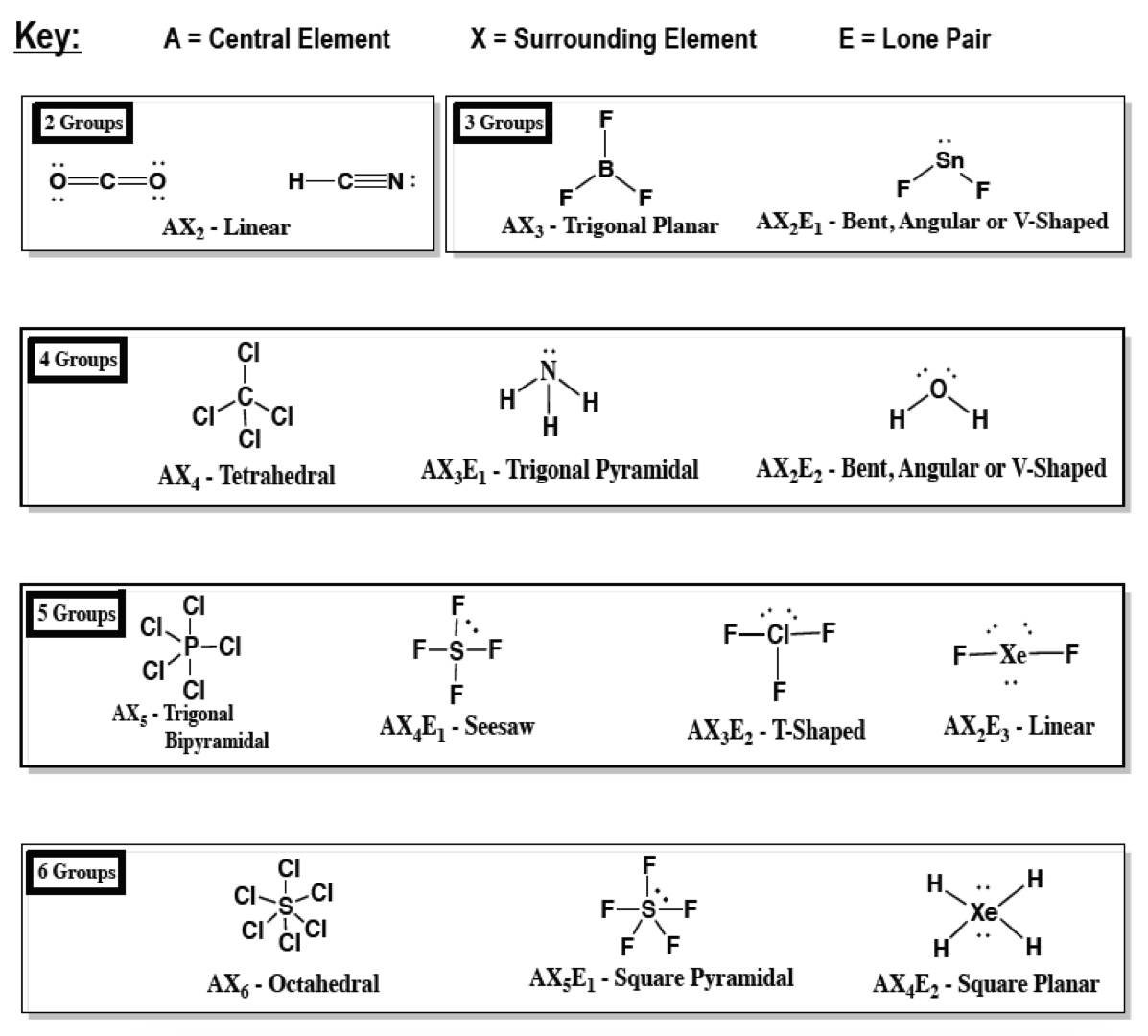
In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. Electron geometry gives us the shape of the molecule that includes both bonding and non-bonding electron pairs on the other hand, molecular geometry gives us the shape of the molecule that includes only the bonding electron pairs.Molecular structure with atoms at the center and vertices of a triangular bipyramid Trigonal bipyramidal molecular geometry The above discussion concludes that both electron geometry and molecular geometry is the geometry that is used for the determination of the shape of the molecule. So, the molecular geometry of the water molecule is bent. It receives two bonding electrons from the hydrogen atom by the sharing method. The central atom is the oxygen molecule having two lone pairs. It contains two bonding electrons and two non-bonding electrons. The example of molecular geometry is the water molecule. The main difference between the electron geometry and molecular geometry is that when we discuss the shape of the molecule, it means we are determining the molecular geometry on the other hand, when we discuss the geometry of electron pairs, it means we are determining the electron geometry.Īs molecular geometry is used to determine the shape of the molecule, so we must use the lewis structure when we are discussing the shape of molecules in the molecular geometry by drawing it in the form of a lewis method to determine the number of bonding electrons. The repulsion between the bonded atoms also considered being less to find out the main purpose of the geometry. It gives us the position of the atoms in the structure of the molecule. In molecular geometry, electron pairs are mainly not considered. Its main purpose is to find out the geometry of the molecule by the arrangement of atoms in a molecule. It is measured by the arrangement of atoms around the nuclei of the central atom. Molecular geometry is the type of geometry that is useful in giving the shape of the molecule that includes only the bond pair of electrons that are present in a molecule. Therefore, it is summarized that the geometry of methane is tetrahedral. There are four single bonds in the structure of the molecule. The four valence electrons are denoted by the hydrogen atom to complete the valence shell of both carbon and hydrogen atoms by the sharing process. The central atom is carbon, and the number of valence electrons is 4. By decreasing the repulsion between these electronic regions, electron geometry gives us the shape of the molecules.Īn example of electron geometry can be given by using the formula of methane. That’s why the electron geometry depends upon the number of these electron regions. Non-bonding electrons are also known as the lone pair of electrons. In electron geometry, it also considers the region of electrons having different densities. 
Electron geometry can also be determined by decreasing the repulsions between the lone pair of electrons. It gives us the arrangement of groups of electrons around the central atom. Electron pairs are considered only in the electron geometry and neglected in the molecular geometry. The main purpose of the electron geometry is to find out the geometry of the molecule by the arrangement of atoms and electrons around the central atom. It can also be determined by the help of the VSEPR Theory. What is Electron Geometry?Įlectron geometry is the type of geometry that is useful in giving the shape of the molecule that includes both electron pairs and bond pairs that are present in a molecule. Electron geometry includes both bonding and non-bonding electron pairs on the other side of the coin, molecular geometry includes only the bonding electron pairs. Electron geometry gives us information about the organization of groups of electrons on the other hand, molecular geometry gives us information about the organization of only atoms in molecule except for lone pairs.Įlectron geometry can be determined by the help of the VESPR Theory on the flip side, molecular geometry is defined by the arrangement of atoms around the nuclei of the central atom. In electron geometry, the total number of pair of electrons are considered on the other side of the coin, in molecular geometry, the total number of electron pairs that are participated in bonding is considered. In electron geometry, lone pairs are also counted on the opposite side, in molecular geometry, lone pairs are not counted. Electron geometry contains electron pairs on the flip side, molecular geometry does not contain electron pairs. Molecular GeometryĮlectron geometry is formed by both the electron pairs and bonds present in a molecule on the other hand, molecular geometry is formed by only bonds that are present in a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
